Monitoring for SARS-CoV-2 in Health Care Facility Environments
Introduction
It is understood that SARS-CoV-2 (as well as influenza and SARS-CoV-1) transmission is primarily through droplet and contaminated surface contact. However, there is significant direct and indirect evidence that aerosol transmission of SARS-CoV-2 is probable when many COVID-19 patients are housed and treated within contained spaces, and when aerosol-generating procedures are used.1-9
In the current reality of the COVID-19 pandemic, hospitals may be housing hundreds of COVID-19 patients and operating blindly when it comes to surface, fomite, and aerosol contamination within COVID-19 wards and within the general hospital.
Traditional methods for aerosol and surface monitoring for bacteria and viruses are negatively affected by limited collection volumes, poor sample recovery, and complex and difficult to perform recovery processes. InnovaPrep offers novel systems that provide simple and straightforward methods for collection and detection of SARS-CoV-2 and other pathogens at previously undetectable levels.
Proactive monitoring provides health care providers with increased understanding of:
- Current state of contamination in the facility
- Effectiveness, limits or gaps of environmental controls
- Effectiveness, limits or gaps of cleaning and disinfection procedures
- Infection risk associated with staffing locations, functions, and available PPE
Enabling introduction of improved control measures and corrective actions and enables PPE use optimization during the current shortage.
Biomonitoring Tools for SARS=CoV-2 in Air, Surfaces, and Liquids
Rapid, easy, small, battery operable and portable devices

Air Sampling – The ADC-200 Bobcat™ is a small lightweight and portable dry filter air sampler with a built-in tripod that actively collects viruses, bacteria and fungal spores from 100 LPM to 200 LPM. Sample recovery from the filter takes just seconds and results in a concentrated liquid sample of 6 mL that is ready for analysis using PCR or other molecular methods.
Longitudinal Metagenomic Analysis of Hospital Air Identifies Clinically Relevant Microbes An article published by the Public Library of Science (PLOS ONE 2016). by Paula King, et al.
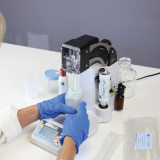
Liquid Concentration for exponentially increasing the limit of detection – the Concentrating Pipette™ will concentrate viruses, bacteria, and fungal spores into a final concentrate of ~200 µL in minutes with the press of a button. The Concentrating Pipette pairs perfectly with PCR and other molecular methods.
Francoise M. Blachere, CDC NIOHS, et al.
A surface sampling method published by NASA JPL showed the effectiveness of how simple felt squares or dry Swiffer-type wipes can be wetted and used to collect samples from large area surfaces. The resulting sample can be concentrated, without enrichment, on the Concentrating Pipette into a PCR-ready sample of about 200 µL.
a poster presentation given by Dave Alburty, InnovaPrep CEO, at the American Society of Microbiology Biothreats meeting in January 2019. The poster outlines three studies where pathogen detection of air, surface and liquid samples were improved by using the Concentrating Pipette for rapid concentration. The studies were performed by independent labs including CDC NIOSH, NASA JPL, and the USDA.

Detection – InnovaPrep elution fluids are compatible with most molecular assays. For those facilities without these capabilities, or those requiring field operation, InnovaPrep’s elution fluids are compatible with Biomeme’s Franklin mobile thermocycler for qPCR and Biomeme’s SARS-CoV-2 Go-Strips assays. Two RNA targets for the novel coronavirus are multiplexed together with Biomeme's process control assay for RNA extraction and RT-PCR (MS2).

About InnovaPrep
InnovaPrep® is a developer of preanalytical tools for modern microbiology. InnovaPrep products provide the critical macro-to-micro interface between real world samples and the input volumes of modern molecular methods for analysis. These tools increase sensitivity and enable a faster, easier, and more efficient means of delivering the most highly concentrated sample possible for subsequent analysis.
InnovaPrep’s 35 pending and awarded patents apply to highly efficient collection and concentration of biological particles from air, surfaces, and liquids. InnovaPrep’s Wet Foam Elution™ process enables instant recovery of particles from filters, membranes, surfaces, and objects. The primary utility for these technologies is to greatly improve the way biological samples, especially dilute samples, are collected and prepared for analysis, allowing the most advanced biological detection systems to contribute their full potential.
___________________________________
1Booth, T. F., et al. (2005). Detection of Airborne Severe Acute Respiratory Syndrome (SARS) Coronavirus and Environmental Contamination in SARS Outbreak Units. The Journal of Infectious Diseases, 191(9), 1472–1477. doi: 10.1086/429634
2Cai, J., Sun, W., Huang, J., Gamber, M., Wu, J., & He, G. (2020). Indirect Virus Transmission in Cluster of COVID-19 Cases, Wenzhou, China, 2020. Emerging Infectious Diseases, 26(6). doi: 10.3201/eid2606.200412
3Doremalen, N. V., et al. (2020). Aerosol and surface stability of HCoV-19 (SARS-CoV-2) compared to SARS-CoV-1. doi: 10.1101/2020.03.09.20033217
4Hugonnet, S., & Pittet, D. (2004). Transmission of Severe Acute Respiratory Syndrome in Critical Care. American Journal of Respiratory and Critical Care Medicine, 169(11), 1177–1178. doi: 10.1164/rccm.2403004
5Ong, S. W. X., et al. (2020). Air, Surface Environmental, and Personal Protective Equipment Contamination by Severe Acute Respiratory Syndrome Coronavirus 2 (SARS-CoV-2) From a Symptomatic Patient. Jama. doi: 10.1001/jama.2020.3227
6Tellier, R., Li, Y., Cowling, B. J., & Tang, J. W. (2019). Recognition of aerosol transmission of infectious agents: a commentary. BMC Infectious Diseases, 19(1). doi: 10.1186/s12879-019-3707-y
7Yu, I. T., et al. (2004). Evidence of Airborne Transmission of the Severe Acute Respiratory Syndrome Virus. New England Journal of Medicine, 350(17), 1731–1739. doi: 10.1056/nejmoa032867
8Giuseppina La Rosa, et al. Viral infections acquired indoors through airborne, droplet or contact transmission. Ann Ist Super Sanità 2013 | Vol. 49, No. 2:124-132
9Santarpia, J. L., et al. (2020). Transmission Potential of SARS-CoV-2 in Viral Shedding Observed at the University of Nebraska Medical Center. doi: 10.1101/2020.03.23.20039446


